Model description
Both capillary and uniform condensation are modeled with the following equation:
where is condensate volume, is area of the vapor-liquid interface, is thermal velocity, is molecular weight of the condensate, is saturated vapor pressure of the condensate, is density of the condensate, is the universal gas constant, is temperature, is vapor saturation, is Kelvin length, is the molecular accommodation coefficient (assumed to be unity here), and is the mean curvature of the vapor-liquid interface. Thermal velocity and Kelvin length are given by:
Note that condensation is considered on a single primary particle and a single junction. Hence, to get quantitative results, calculated capillary and uniform coating volumes need to be multiplied by the number of primary particles present in the aggregate of interest.
The integration time step is computed based on the characteristic condensation rate:
where is a constant equal to . Hence, the time step is inversely proportional to the characteristic condensation rate and is larger for less volatile materials. Thus, the residence time must be increased when modeling less volatile coatings to ensure that there are enough points to resolve the filling angle vs. time profile.
Coating geometry
If vapor is supersaturated (), exceeding a critical value necessary to overcome the Kelvin effect, it can condense on the convex surfaces of the spherical primary particles in an aggregate, thus engaging in “uniform condensation”. Otherwise, condensation is only possible on the concave surfaces between the primary particles. Condensation in those gaps is called “capillary condensation”.
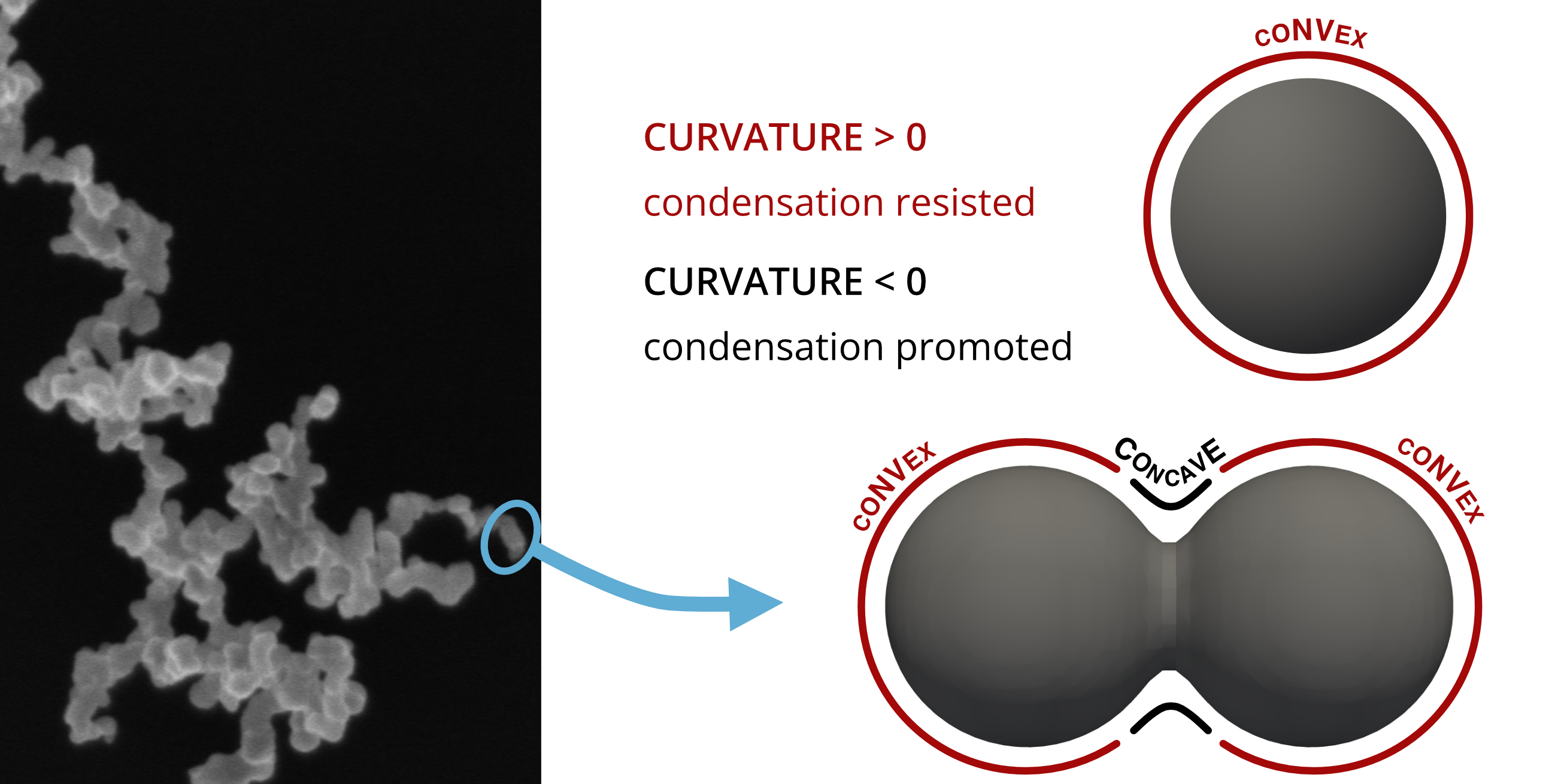
The mode of condensation that will occur in a given system can be estimated using the parameter proposed by Chen et al. 1:
If the parameter is close to unity, capillary condensation is expected. If is much lower than one, then both capillary and uniform condensation will occur.

The model implemented in this web application considers both possible condensation modes. For the purpose of uniform condensation, condensate on the primary particles is assumed to have a core-shell geometry, and the corresponding area and curvature are used. Geometry of the capillary condensate is more complicated; As described by Ivanova et al. 2, condensate in a gap between two primary particles is assumed to be a body with a surface of constant mean curvature. The liquid profile can be obtained by solving the following differential equation:
where is mean curvature of the revolution surface non-dimensionalized with the primary particle radius, . The equation becomes a boundary value problem with an unknown parameter, thus requiring three boundary conditions, which are:
where is the contact angle of the coating material. Volume and surface area of the revolution surface can be computed numerically from the solution. In practice, a table of curvatures, volumes, and surface areas of the constant mean curvature surface is pre-computed for a range of and combinations. Curvatures and areas are then interpolated between available points and scaled by the primary particle radius when solving the condensation equation.
Vapor saturation
Vapor saturation is the main parameter controlling the mode and rate of condensation. The magnitude of saturation depends on the source of condensable vapor. Typically, vapor becomes supersaturated () either when it is suddenly cooled or when its volatility is reduced through a chemical reaction. Otherwise, if vapor is in equilibrium with a sink, it is usually subsaturated (). Thus, methods to estimate saturation will vary based on the system of interest. For example, in a laboratory study of capillary condensation by Khalizov et al. 3, steady-state saturation of triethylene glycol (TEG) in a partially filled tube at room temperature was estimated with finite element modeling to be 0.9.
Preset compounds
Preset compounds available in this application are triethylene glycol (TEG), dioctyl sebacate (DOS), and 78% by weight aqueous solution of sulfuric acid (SA). Additionally, some condensable compounds used in the MOSAIC atmospheric chemistry simulator 4 were added as presets. These are lumped compounds representing oxidation products of α-pinene (API1, API2), limonene (LIM1, LIM2), and aromatics (ARO1, ARO2) 5.
Technical requirements
A modern web browser with WebAssembly and web worker support and JavaScript enabled is required to run the application. The application should work on both desktop and mobile devices, as long as the web browser is up to date. The application was tested in Safari, Firefox, and Chrome. Note that calculations run on the client side, thus performance will vary between devices. Please, report bugs and suggestions to Egor Demidov at ed242@njit.edu.
Code availability
The capillary condensation model, including support for multi-component systems, was implemented in an open source C++ library. This web application was built with the C++ library compiled into web assembly, which allows C++ code to run in modern web browsers with near-native performance.
Users of this model
Escritt et al. 6 used the capillary condensation model to interpret their laboratory data, showing that coating material preferentially accumulates at interparticle contact points, enabling efficient restructuring of black carbon at extremely low coating volumes.
Khalizov et al. 3 used the offline version of the capillary condensation model, coupled with a particle-resolved aerosol model, to explain rapid soot restructuring and aging under atmospherically relevant conditions.
Citing this work
If you find the capillary condensation model useful for your research, please cite the following paper to acknowledge the use:
@article{khalizov2025capillary,
title={Capillary Condensation: An Unaccounted Pathway for Rapid Aging of Atmospheric Soot},
author={Khalizov, Alexei F and Ivanova, Ella V and Demidov, Egor V and Hasani, Ali and Curtis, Jeffrey H and Riemer, Nicole and Gor, Gennady Y},
journal={Environmental Science \& Technology},
volume={59},
number={28},
pages={14564--14571},
year={2025},
publisher={ACS Publications}
}
Footnotes
-
Chen, C., Enekwizu, O. Y., Fan, X., Dobrzanski, C. D., Ivanova, E. V., Ma, Y., Gor, G. Y., & Khalizov, A. F. (2018). Single parameter for predicting the morphology of atmospheric black carbon. Environmental Science & Technology, 52(24), 14169-14179. ↩
-
Ivanova, E. V., Khalizov, A. F., & Gor, G. Y. (2020). Kinetic model for competitive condensation of vapor between concave and convex surfaces in a soot aggregate. Aerosol Science and Technology, 55(3), 302-315. ↩
-
Khalizov, A. F., Ivanova, E. V., Demidov, E. V., Hasani, A., Curtis, J. H., Riemer, N., & Gor, G. Y. (2025). Capillary Condensation: An Unaccounted Pathway for Rapid Aging of Atmospheric Soot. Environmental Science & Technology, 59(28), 14564-14571. ↩ ↩2
-
Zaveri, R. A., Easter, R. C., Fast, J. D., & Peters, L. K. (2008). Model for simulating aerosol interactions and chemistry (MOSAIC). Journal of Geophysical Research: Atmospheres, 113(D13). ↩
-
Schell, B., Ackermann, I. J., Hass, H., Binkowski, F. S., & Ebel, A. (2001). Modeling the formation of secondary organic aerosol within a comprehensive air quality model system. Journal of Geophysical Research: Atmospheres, 106(D22), 28275-28293. ↩
-
Escritt, C. A., Al-Mashala, H. H., & Schnitzler, E. G. (2026). Geranyl ester components of volatile chemical products readily induce black carbon restructuring. Environmental Science: Processes & Impacts. ↩